Strategic opening in the Pharmaceutical Sector
Repeal of the 2012 Patentability guidelines and new regulatory framework
1. Context of the News
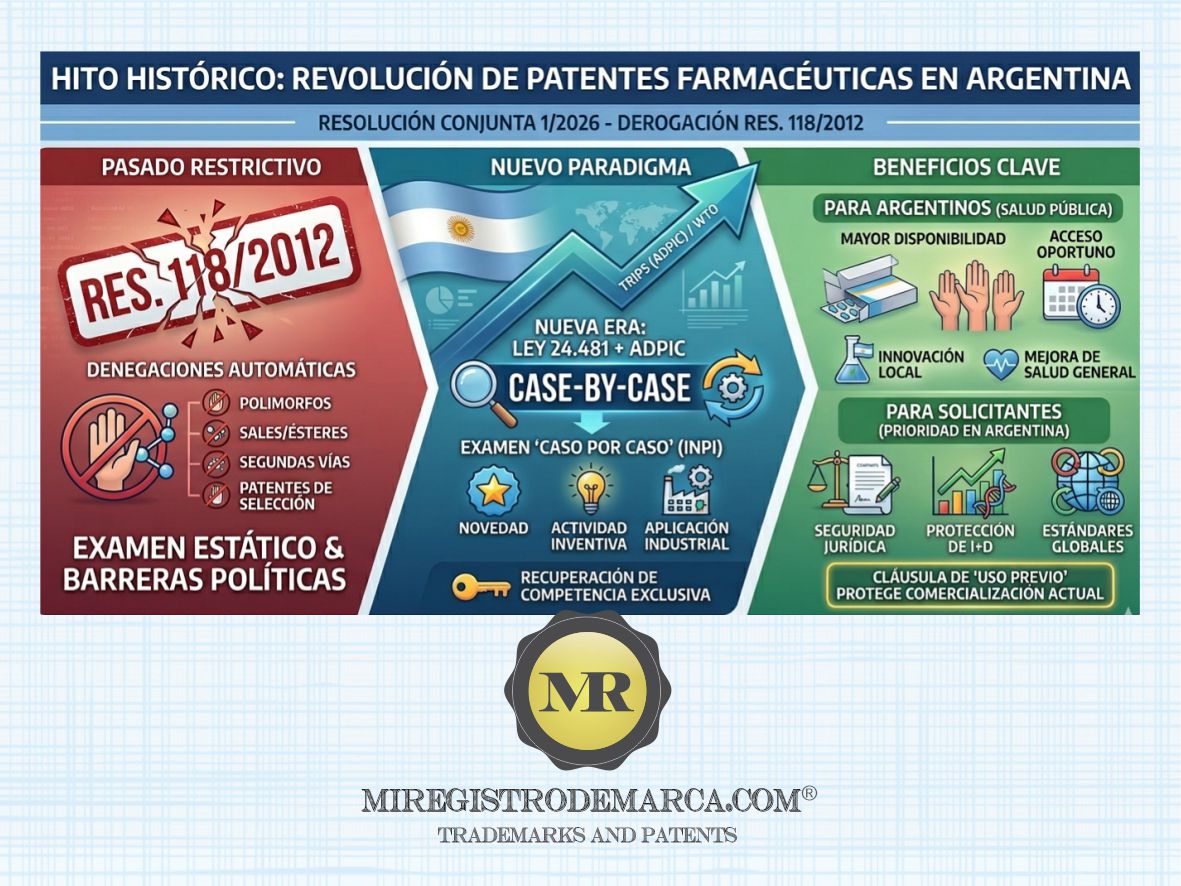
On March 17, 2026, through Joint Resolution 1/2026 (Health, Economy, and INPI), the National Government ordered the total repeal of Joint Resolution 118/2012. The latter was known in the sector for imposing strict and restrictive criteria on the examination of chemical-pharmaceutical patent applications, which often limited the protection of incremental innovations.
2. Key points of the resolution
A. End of automatic restrictions With the elimination of the 2012 guidelines, the INPI regains its exclusive competence to analyze each application on a "case-by-case" basis. There will no longer be presumptions of lack of novelty or inventive step based solely on the category of the invention (such as polymorphs, salts, esters, or processes).
B. Alignment with the TRIPS agreement (WTO) The regulation bases its decision on Article 27.1 of TRIPS, which prohibits discrimination by field of technology. The objective is to:
- Foster local and international innovation.
- Increase the availability of new drugs in the Argentine market.
- Guarantee legal certainty and predictability for R&D investments.
C. Safeguard Mechanism (Transitional Clause) To prevent the granting of new patents (previously deniable under the old guidelines) from disrupting the current supply of medicines, Article 2 establishes a limited exception:
- Scope: Patents granted as of this resolution.
- Protection: If a third party is already locally marketing a pharmaceutical product linked to that new patent at the time of enactment, the patent holder may not prevent the third party from continuing its commercialization.
- Compensation: The holder shall not have the right to demand remuneration or royalties for such pre-existing commercialization.
3. Impact matrix for Professionals and Companies
| Area | Main impact | Strategic consideration |
| R&D / Pipeline | Higher probability of grants for incremental inventions. | Review pending files and reactivate protection strategies. |
| Legal / Litigation | New interpretation of patentability requirements (Law 24.481). | Examination returns to being purely technical-legal according to the Patent Law. |
| Commercial | Faster entry of innovative drugs into the local market. | Monitor the state of the art and competitors' applications. |
4. Technical Conclusion
Resolution 1/2026 eliminates the regulatory barriers that differentiated the pharmaceutical sector from other technological fields. By adopting World Trade Organization (WTO) standards, Argentina seeks to position itself as an attractive destination for the simultaneous launch of next-generation medicines, balancing this right with a "prior use" clause that protects the stability of current market players.
Recommended next steps As a professional in the field, the coming days will be crucial for:
- Portfolio Audit: Identify applications that were subject to office actions or objections based specifically on Res. 118/2012 (now repealed)
- Preparation of Arguments: Adjust responses to legal notices by focusing exclusively on Novelty, Inventive Step, and Industrial Application.
Comparative matrix of patentability criteria
| Technical matter | Repealed regime (Res. 118/12) | New regime (Res. 1/2026 + Law 24.481) |
| General principle | Restrictive: Presumption of lack of inventive step for incremental innovations. | Technical Sovereignty: "Case-by-case" analysis strictly based on Novelty, IS, and Industrial App. |
| Polymorphs | Considered "discoveries" (non-patentable). Required proof of extreme unexpected effects. | Patentable: If the polymorph is new and not obvious to an expert in the field. |
| Salts, Esters, and Ethers | Considered the same active substance. Systematically denied except for rare exceptions. | Patentable: Evaluation of whether the new chemical form provides a real inventive step over the base. |
| Selection patents | Practically prohibited. Disclosures in a general formula were deemed to kill novelty. | Accepted: Following international doctrine, if the selection is not arbitrary and offers an unsuggested technical advantage. |
| Formulations and compositions | Required to be "non-obvious" and solve a problem in a surprising way. | Law criterion: The general standard of inventive step (Art. 4) applies. |
| Second Pathways/Processes | Very limited. Changes in manufacturing processes were usually considered obvious. | Process protection: The value of innovation in manufacturing methods is recognized (Art. 4 and 10). |
Change in the Burden of Proof and Procedure
Before (Under Res. 118/12): The examiner used the "Guidelines" as an entry barrier. If an application fell into a category such as "Polymorphs," the burden of proof for the applicant was almost impossible to satisfy, as the regulation pre-established that they were non-patentable.
Now (Under Res. 1/2026): Technological Neutrality: The examiner cannot prejudge the invention for being pharmaceutical.
- INPI autonomy: External political directives (Ministry of Health and Industry) regarding the patent examiner's technical criteria are eliminated.
- Jurisprudence and doctrine: The value of Law 24.481 and the TRIPS Agreement are recovered as the sole frames of reference.
The "Prior Use" clause (Art. 2)
It is fundamental to distinguish that, although patents previously denied may now be granted, there is a limit to the exercise of the right:
Golden rule: If you have a client who already sells a generic or product whose patent is granted NOW due to this change, the holder of that new patent cannot sue them or demand royalties.
This is a direct application of Art. 41 of the Patent Law and Art. 30 of TRIPS, which allows limited exceptions to protect the legal certainty of those who invested under the previous regime.
Impact on Prosecution Strategy
For applications currently on the examiner's desk, it is recommended to:
- File amendments: If claims had been limited to attempt to circumvent Res. 118/12, evaluate the possibility of broadening them again (provided they have support in the original description).
- New arguments: Cite Res. 1/2026 in responses to office actions to invalidate any objection based on the repealed guidelines.
Read Joint Resolution 1/2026 RESFC-2026-1-APN-INPI#MEC, published in the official gazette of the Argentine Republic.